Rhino Husbandry Manual

White Rhinoceros
Behavior
Ranging Behavior and Sociality
Females and Subadults
The behavioral ecology of the social white rhinoceros differs from that of the other rhino species, which tend to be more solitary (Owen-Smith, 1988; Hutchins and Kreger, 2006). White rhinos have a rich social repertoire, displaying a variety of behaviors and vocalizations (Table 2.24) regularly during interactions. Pairs or small groups of white rhino females and adolescents are the norm (Owen-Smith, 1973, 1975; Pienaar, 1994; Shrader and Owen-Smith, 2002), although most of these associations are transient, lasting only a few months (Owen-Smith, 1973, 1975; Shrader and Owen-Smith, 2002). Close companions are typically two animals, such as mother and calf, two adolescents, or an adolescent and an adult female; only rarely are two adult females found in long-lasting associations in the wild (Owen-Smith, 1973; Hillman-Smith, 1987; Shrader and Owen-Smith, 2002). In contrast, in zoos, companion subgroups can be composed of only adult females (Kuneš and Bičík, 2002; Swaisgood et al., 2006; Metrione et al., 2007). There is some anecdotal evidence that groupings can be comprised of the same pairings listed for their wild counterparts in the previous sentence in addition to adult females. (Cseplo; 2023) The space between companions is usually less than 5 m (16.4 ft) and rarely more than 25 m (82 ft) (Owen-Smith, 1973; Metrione 2005), and aggressive interactions between close companions are exceedingly rare.
Female white rhinos and any social partners accompanying them occupy large home ranges in the wild (6.5-66.8 km2/1,606-16,507 acres; Pienaar, 1994; White et al., 2007a), which overlap extensively with those of other female groups, but the majority of activity may be in smaller core areas about a quarter of the size of the home range (Owen-Smith, 1973, 1975; Pienaar, 1994; White et al., 2007a). Core area use may also shift seasonally, presumably to track resources. Home range area is also influenced by social density, with females ranging farther at lower densities (Owen-Smith, 1975; Rachlow et al., 1999; White et al., 2007a), suggesting that even the relatively gregarious white rhino female limits its movements to avoid social encounters with less familiar rhinos. Female home ranges are substantially larger than male territories, which are socially constrained, with the average female range overlapping with seven male territories (range: 4-15; White et al., 2007a). While extensive space might be available at some zoological institutions for rhinos, an entire group, subdivided into companion subgroups, often can be found in the same general area of a large pasture (Metrione et al., 2007), consistent with the temporary social aggregations seen in wild rhino. Some analyses indicate that larger enclosures are associated with improved reproduction (Swaisgood et al., 2006; Metrione, 2010).
As noted above, associations between free-ranging individuals, particularly those that involve adolescents, frequently last less than a month, but some may persist for five or more months (Owen-Smith, 1973, 1975; Shrader and Owen-Smith, 2002). Relationships in zoological institutions can be long-lasting (years) and might change only when a calf is born to one of the companions (Metrione et al., 2007). In fact, female white rhinos housed with a female companion known from adolescence tended to have a lower average concentration of corticosterone, a hormone associated with the stress response, than females housed with recently-introduced female companions or no female companion (Metrione and Harder, 2011). In addition, in managed breeding settings, better reproduction is associated with housing females in groups of three or more females/adolescents (Metrione, 2010), including housing captive-born females with wild-caught females (Swaisgood et al., 2006).
Dominance hierarchies probably do not occur in the wild (Owen-Smith, 1973), but in the zoo setting, dominant females can gain access to food sooner and feed for longer durations than subordinate females when the group is fed in specific and spatially restricted areas (Metrione et al., 2007). Aggression and the frequency of space-maintenance vocalizations, such as the snort and snarl (Owen-Smith, 1975; Table 2.24), usually are higher during these feeding times than at other times during the day (Metrione et al., 2007). Positive contact behaviors, such as play, might help to mitigate any distress caused by aggressive interactions, especially for subordinate females (Metrione and Harder, 2011). Excessive horn rubbing (Hutchins and Kreger, 2006; Table 2.24), on the other hand, is a behavior that might indicate boredom or distress. Higher rates of stereotypic behavior were reported for non-cycling female white rhinos compared with cycling females (Carlstead and Brown, 2005).
Males
Reproductive white rhino males, which are only 33 to 67% of all wild adult males, establish and defend non-overlapping territories approximately 5 km2 (1,235 acres) in size, while non-reproductive, subordinate males live within a dominant male’s territory (Owen-Smith, 1971, 1973, 1975; Rachlow, 1997; Rachlow et al., 1999; White et al., 2007a). Territory boundaries are delineated by scent marks left only by territorial males during spray-urination, dung-kicking, scraping with the fore- and/or hindlegs, and horn-scraping (Table 2.24; Owen-Smith, 1973, 1975). These behaviors also are expressed by zoo-managed white rhino males throughout their enclosures. When wild territorial male rhinos temporarily move off of their territory in search of water during drought, they adopt the behavior of a subordinate male (e.g., urinating in a stream as opposed to spraying urine), even in the presence of a female (Owen-Smith, 1973). Subordinate males rarely associate with estrous females for any significant length of time, while territorial males associate with a significantly larger number of such females (Rachlow, 1997; Rachlow et al., 1998).
Confrontations between territorial males are highly ritualized, involving advancements, retreats, horn-scraping, horn-clashes directed mainly at the opponent’s horn, and checked jabbing gestures as opposed to injury-causing movements (Table 2.24; Owen-Smith, 1973, 1975). Some confrontations, however, particularly during a territory take-over, might be prolonged and bloody (Owen-Smith, 1973, 1975), corresponding with Level 5 aggression in Table 2.25. Most wild males, therefore, will not become territory holders until they are 12 years of age or older (Owen-Smith, 1973). The average duration of occupation of the same territory by a male was 5.4 years, after which he might continue to be a territorial male in a different territory (Owen-Smith, 1973).
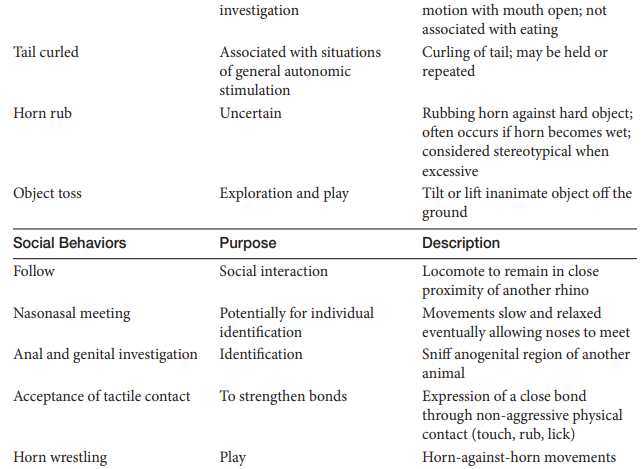
White Rhinoceros Ethogram
In order to conduct appropriate behavioral studies, many institutions have utilized ZooMonitor.
-
ZooMonitor is an app developed by Lincoln Park Zoo that allows you to record animal behavioral data, visualize the information, and export the data for analysis. When designing ZooMonitor, our goal was to make it easy for experts and non-experts alike to collect data that will aid in making and evaluating management decisions that impact animal welfare.
-
ZooMonitor was made possible in part by the Institute of Museum and Library Services MG-30-14-0045-14, and is free to download for qualifying institutions.

Heading 6


Reproductive Behavior
Both in the wild and in managed settings, male and female rhinos defecate almost exclusively at communal dung piles and often spend time investigating excrement at the dung piles (Table 2.24; Owen-Smith, 1973; Metrione, 2005). Female rhinos often urinate at dung piles as well. It has been suggested that the dung piles serve as a “bulletin board” (Owen-Smith, 1973). Both urine and dung serve as chemical signals, and experiments have shown that rhinos can discriminate sex, reproductive condition and individual identity, and they gain other information from these odor sources (Swaisgood, unpublished data). Probably by identifying cues of upcoming sexual receptivity in female excreta, males have been observed intensively following a female as much as a week prior to the onset of estrus (Owen-Smith, 1973). Anestrous female white rhinos are intolerant of a male’s presence and will show aggression to males within 10 m (30 ft) in the wild (Owen-Smith, 1973, 1975). In zoos, the acceptable male-female inter-individual distance seems to vary from rhino to rhino. Male aggression towards females is rare, occurring less than 1/100th as commonly as affiliative behavior toward the female (Swaisgood et al., 2006). The female shows substantially less affiliative behavior toward the male than vice-versa and more aggression, although rates are still low. The pre-estrus consort period lasts up to 20 days in the wild, during which the territorial male is generally obedient to the space-maintenance threats of the female, unless she approaches the border of his territory, upon which the male will approach, vocalize, and charge the female to keep her from leaving (Owen-Smith, 1973). Prior to full estrus, females will use mock charges and defensive threats to drive a bull back (Fouraker and Wagener, 1996). In the case of zoo-managed rhinos, the bull may attempt to split female groupings apart during courtship and breeding attempts. In some cases, these bonds may interfere with breeding success if the females are able to continually drive-off an approaching male (Fouraker and Wagener, 1996).
Mating is a long, slow process. Estrus typically lasts for 24 hours (Owen-Smith, 1973, 1975; Metrione, 2005), during which the male makes regular advances and “hiccing” vocalizations (Table 2.24; Owen-Smith, 1973) and unfailingly makes olfactory investigations of the squirts of urine released by the female (Tables 2.24 and 2.26; Fig. 2.12; Owen-Smith, 1973, 1975; Kuneš and Bičík, 2002; Metrione, 2005). Eventually, the female tolerates chin-resting by the male, usually on her hindquarters or back (Fig. 2.13), mounting attempts (Fig. 2.14), and finally copulation (Tables 2.24 and 2.26; Owen-Smith, 1973, 1975; Swaisgood et al., 2006). White rhino males often mount before an erection occurs (Fouraker and Wagener, 1996). Copulation lasts 15-30 minutes and might include multiple ejaculations (Owen-Smith, 1973, 1975; Swaisgood et al., 2006). It is a good idea to keep a record of all observations of sexual behavior, and suggested instructions for doing so, as well as a data sheet, are provided in Appendices E and F, respectively.
Table 2.26 General reproductive behaviors observed during estrus and courtship (Fouraker and Wagener, 1996)
Female Behaviors Male Behaviors
Vocalizations* Vocalizations
Urine squirt/spray* Frequent urination; urine spray
Urogenital changes (e.g., vulva swelling) Erection
Vulva “winking” Genital inspection of female
Vaginal discharge* Flehmen response
Aggression toward male** Charge/chase female
Maintains proximity to male Maintains proximity to female
Nuzzles male’s belly and/or genitals Follows female
Stands for male Chin-rest
Inappetance Mounts female
*Estrous behaviors in the absence of a male are often difficult to distinguish. In general, increased activity, agitation, vocalizations, spray-squirting urine, and vaginal discharge have been cited. As the female approaches peak estrus, these behaviors usually increase in frequency. Some females have been reported to successfully breed without exhibiting any overt behavioral signs of estrus
**This may be applicable to early stages of estrous.
There is no evidence for the suppression of sexual behavior in captive-born females by wild caught females, including the mother, and copulatory behavior is not compromised in captive-born females (Swaisgood et al., 2006). There also is no evidence that reproduction is influenced by social dominance (Swaisgood et al., 2006; Metrione and Harder, 2011). White rhino males are polygynous (Owen-Smith, 1973), and males were equally likely to direct courtship behaviors toward wild caught and captive-born females during peak estrus (Swaisgood et al., 2006). Among females that have copulated, captive-born females are significantly less likely to give birth, even when years of opportunity is held constant (Swaisgood et al., 2006). These results indicate that the captive-born reproduction problem is post-copulatory, and there must be problems with conception or pregnancy maintenance (Swaisgood et al., 2006; Metrione, 2010). While companions may accompany adult females with older calves, females do not tolerate companions when they are accompanied by a young calf (Owen-Smith, 1973; Shrader and Owen Smith, 2002; Metrione et al., 2007). In fact, females seek dense brush in areas not frequented by other rhinoceros to give birth in the wild (Owen-Smith, 1973) and sometimes will separate from the rest of the group approximately 20 hours prior to parturition in the zoo setting (Metrione et al., 2007). Parturition usually lasts ten to 12 hours from water-break, though first-time mothers may take longer to calve (Fouraker and Wagener, 1996).

Fig. 2.12. Many, but not all, female white rhinos squirt urine and have vaginal discharge during estrus (photo taken at Wildlife Safari)
Maternal care is also highly developed in white rhinos, and the mother-calf bond is undoubtedly the strongest social association in the species (Owen-Smith, 1975; White et al., 2007b). Mothers respond to solicitation vocalizations by calves by cessation of activity and repositioning to allow nursing. Young calves in the wild suckle almost once an hour for about three minutes, spending about 4% of their time suckling (White et al., 2007b). These values decrease as the calf ages, and weaning occurs at about 27 months for males and 19 months for females. Female mothers in good condition invest more resources in male than in female calves (White et al., 2007b). In the managed setting, if the dam and calf were separated from the group, a dam and her newborn calf may be reintroduced to the male/herd after two weeks (Fouraker and Wagener, 1996).
Adolescent White Rhinos
Adolescent dispersal in the wild is instigated by the mother’s aggression at the birth of her next calf (Owen-Smith, 1973). In zoos and conservation centers, adolescent females might remain close companions with their mothers if a new calf is not born to either one. Owen-Smith (1973) observed that male adolescents were sometimes chased by territorial males. Regardless of sex, excursions from home ranges usually occur in pairs or small groups, either with another adolescent(s) or an unrelated adult female (Owen-Smith, 1973, 1975; Shrader and Owen-Smith, 2002).
Behavior During Introductions (from Fouraker and Wagener, 1996)
As noted above, white rhinos in stable groups are highly gregarious and aggression is rare, although there is some variability across individuals and facilities. By contrast, introductions of unfamiliar animals to the group often result in aggression, and it should be noted that rhinos of both sexes have been the aggressors. Behaviors that have been noted during rhino introductions are listed in Table 2.27. Territorial defense is often limited to ritualized confrontations, in which two rhinos advance toward each other but stop and engage in a horn-to-horn stare (Tables 2.24 and 2.27). Also as part of this ritual, the two individuals may touch horns, back apart, and wipe their horns on the ground (Table 2.24; Nowak, 1991). More intensive conflicts (Table 2.25) involve charges and inflicting injuries by horning or charging. It is important to note that what is often perceived as serious or dangerous aggression between rhinos is, in fact, normal behavior requiring no intervention of any kind. Along with increased size and thick skin comes decreased vulnerability compared with many other animals.
In some cases, however, aggression may proceed to a point at which management should intervene to prevent serious injury. Managers should allow some aggression during an introduction but be prepared to intervene in the event that aggression threatens the lives of one or more rhinos. Protocols for intervening may vary across institutions, but in general, careful consideration should be given to intervening in an introduction before aggression reaches Level 5 (Table 2.25). Stopping an introduction at a level prior to this will not lessen aggression during a subsequent introduction attempt. Animals that are allowed to “settle their differences” will establish some territorial boundaries and will usually not engage in serious aggression again, with the exception of a male attempting to approach an estrous female. In sum, moderate aggression is commonplace in any rhino introduction; sparring and fighting will occur and result in minor injuries (e.g., cutaneous wounds). However, in most cases, aggression levels prior to Level 5 may be allowed to continue using the discretion of management.
Table 2.27 Behaviors noted during rhino introductions (Fouraker and Wagener, 1996).
Non-aggressive Behaviors Ritualized Confrontations Potential Stress-Related Aggressive Behaviors
Follow Head sweep Pacing Charge/chase
Touch/rub/lick Face-to-face stare Running (excessive) Open-mouth threat
Anal or genital Space-maintenance and Sparring
investigation threat vocalizations Goring
(excessive)
Diarrhea
Animal personality and disposition should always be considered in introductions. A subordinate animal should be introduced to a more dominant animal in an enclosure familiar to the subordinate. In the case of multiple-animal introductions (e.g., introduction of a new female to an established male-female group), the most subordinate animal should be introduced to the next most subordinate, and so on up the dominance hierarchy. Greater aggression may be noted in some individuals in the presence of an estrous female; therefore, any introduction attempt at this time should be especially well-monitored or possibly avoided if the attempt involves a male. Aggression may be meaningfully reduced by allowing individuals to become familiar with each other through protected contact (e.g., through enclosure bars) or, possibly, by exposing them to one another’s odors for a period prior to introduction.

Fig. 2.14. The male usually mounts the female multiple times before successful copulation finally occurs (photo taken at Wildlife Safari).